Cefcum s 750
Price 125 INR/ Case
Cefcum s 750 Specification
- Pacakaging (Quantity Per Box)
- Each box contains 1 vial
- Salt Composition
- Ceftriaxone Sodium
- Indication
- Treatment of infections caused by susceptible organisms
- Origin of Medicine
- Allopathic
- Dosage Form
- Injection
- Drug Type
- Prescription
- Ingredients
- Ceftriaxone Sodium 750 mg
- Physical Form
- Powder for Injection
- Function
- Antibiotic
- Recommended For
- Bacterial Infections
- Dosage
- As directed by physician
- Dosage Guidelines
- Intravenous (IV) or Intramuscular (IM) use only
- Suitable For
- Adults & Pediatric
- Quantity
- 1 vial
- Storage Instructions
- Store below 25C, protected from light and moisture
- Therapeutic Class
- Third Generation Cephalosporin
- Shelf Life
- 24 months from manufacturing date
- Prescription Required
- Yes
- Marketed By
- Cumbus Pharmaceuticals
- Brand Name
- Cefcum s 750
- Route of Administration
- IV/IM
- Pack Size
- Single vial in box
- Regulatory Approval
- WHO GMP Certified
- Strength
- 750 mg
- Colour
- White to off-white powder
- Usage
- Hospital and clinical use
- Reconstitution
- Reconstitute with sterile water for injections
- Contraindications
- Known hypersensitivity to cephalosporins
Cefcum s 750 Trade Information
- Minimum Order Quantity
- 100 Cases
- Main Domestic Market
- All India
About Cefcum s 750
Ceftriaxone+Sulbactam is used in the treatment of bacterial infections. Ceftriaxone + Sulbactam is a combination of two medicines: Ceftriaxone and Sulbactam. Ceftriaxone is an antibiotic. It works by preventing the formation of the bacterial protective covering which is essential for the survival of bacteria.Potent Antibiotic Therapy
Cefcum s 750 offers broad-spectrum antibacterial action as a third-generation cephalosporin. Its efficacy covers diverse bacterial pathogens, making it a reliable choice for hospitals and clinics treating serious infections. Each vial contains sterile Ceftriaxone Sodium, intended for rapid clinical effect and patient recovery.
Usage and Administration Guidelines
Cefcum s 750 is administered intravenously or intramuscularly. The powder form must be reconstituted with sterile water before injection. Dosages depend on infection type and patient profile, and should always follow the physician's guidance. Suitable for both adults and children, it is available as a single vial in secure packaging.
Quality Assurance and Certification
Produced by Cumbus Pharmaceuticals, Cefcum s 750 is WHO GMP certified, ensuring its safety, quality, and effectiveness in clinical practice. With a 24-month shelf-life and robust packaging, it is designed to maintain potency during storage and transport to global healthcare facilities.
FAQ's of Cefcum s 750:
Q: How should Cefcum s 750 be administered?
A: Cefcum s 750 is intended for intravenous (IV) or intramuscular (IM) injection only. The powder should be reconstituted with sterile water for injection immediately before administration as directed by a healthcare professional.Q: What infections can be treated with Cefcum s 750?
A: Cefcum s 750 is recommended for the treatment of infections caused by susceptible bacteria, including respiratory tract, urinary tract, abdominal, skin, and soft tissue infections. Your physician will decide its suitability based on diagnosis.Q: When should I avoid using Cefcum s 750?
A: Do not use Cefcum s 750 if you have a known hypersensitivity to cephalosporins or any component of the formulation. Always inform your physician about any allergies prior to receiving this medication.Q: Where should Cefcum s 750 be stored?
A: Store Cefcum s 750 below 25C, protected from light and moisture. Proper storage helps in maintaining its efficacy throughout the 24-month shelf-life from the manufacturing date.Q: What is the process for preparing Cefcum s 750 for injection?
A: To prepare Cefcum s 750, add the prescribed amount of sterile water for injection to the vial and shake gently until the powder dissolves completely. The solution should be used immediately after reconstitution.Q: What are the benefits of using Cefcum s 750?
A: Cefcum s 750 provides effective, broad-spectrum coverage against a variety of bacterial infections. Its powerful action and validated manufacturing standards contribute to reliable patient outcomes in medical settings.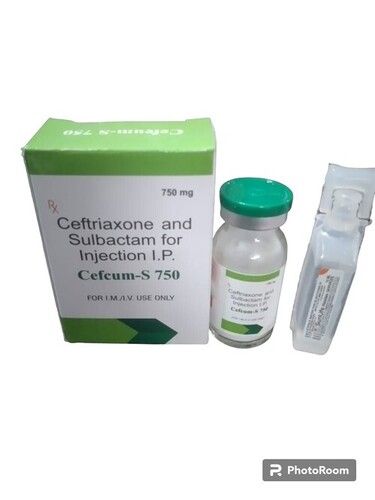

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Injectables Category
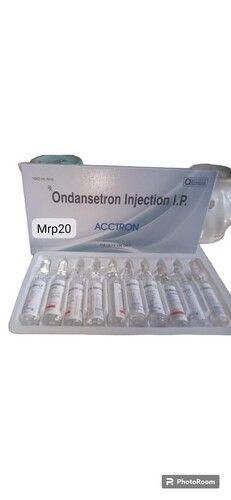
Acctron injection
Price 20 INR / Case
Minimum Order Quantity : 100 Cases
Indication : Respiratory tract infections, Urinary tract infections, Skin and soft tissue infections, Intraabdominal infections, Bone and joint infections, Septicemia, Meningitis, Gonorrhea
Recommended For : Bacterial Infections
Drug Type : Other, Prescription
Origin of Medicine : Allopathic
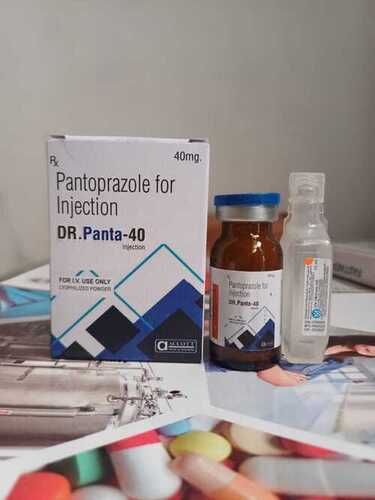
DR PAnta 40 Injection
Price 49 INR / Piece
Minimum Order Quantity : 100 Pieces
Indication : Gastric ulcers, duodenal ulcers, ZollingerEllison syndrome, GERD, erosive esophagitis
Recommended For : Adults and children as prescribed
Drug Type : Other, Proton Pump Inhibitor (PPI)
Origin of Medicine : Allopathic
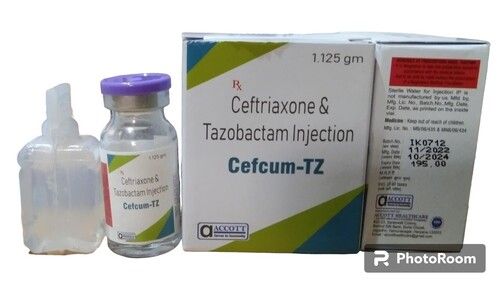
Cefcum TZ
Price 195 INR / Case
Minimum Order Quantity : 100 Cases
Indication : Treatment of bacterial infections
Recommended For : Bacterial infections
Drug Type : Other, Antibiotic
Origin of Medicine : Allopathic
Redflam 75 (Redflam)
Price 19 INR / Case
Minimum Order Quantity : 100 Cases
Indication : Rheumatoid arthritis, osteoarthritis, ankylosing spondylitis
Recommended For : Pain relief, inflammations associated with arthritis and musculoskeletal disorders
Drug Type : Other, Nonsteroidal antiinflammatory drug (NSAID)
Origin of Medicine : Allopathic


 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS